Ock-Joo Kim*+., Byung-Joo Park** +, Seung-Mi Lee**, Dong-Ryul Sohn,*** Sang-Goo Shin**** + Department of Medical Education Korea University College of Medicine*, Department of Preventive Medicine Seoul National University College of Medicine**, Department of Clinical Pharmacology Soonchunhyang University College of Medicine***, Department of Pharmacology Seoul National University College of Medicine and Clinical Pharmacology Unit/SNUH****, The Korean Association of Institutional Review Boards (KAIRB) + Address for Correspondence: Sang-Goo Shin, M.D., Ph.D. Department of Pharmacology Seoul National University College of Medicine and Clinical Pharmacology Unit/SNUH, 28 Yeongon-dong Chongno-gu, Seoul 110-799, Korea E-mail:capmed01@snu.ac.kr
pp.17-27 in Song, SY, Koo, YM & Macer, DRJ. eds.
Bioethics in Asia in the 21st Century (Eubios Ethics Institute, 2003).
The Declaration of Helsinki uniformly requires all biomedical research involving human participants including research on identifiable human material or data should be approved by ethical review committee (1). As it evolved out of the scandalous unethical research practice in the late twentieth century, ethics review of study protocols by independent ethics committee (IEC) or institutional review boards (IRBs) has become an international standard of ethically and scientifically acceptable biomedical research.
Today, concerns over the quality of IRB functions are increasing worldwide. As globalization of the clinical trials in the last decade has led to more people in developing countries participating in the trial, international efforts to assure the participant's right and safety, such as producing more guidelines and regulations or organizing conferences on ethics reviews, have been made recently. Nevertheless, voices of worrying over the protections of research participants have been growing among researchers of developing countries as well as oversight bodies in developed countries. Most of all, the IRB function to ensure quality of research in scientific and ethical aspects differs from place to place for various reasons. In spite of growing importance of ethics review on human participant research, concrete information on the performance of IRBs are not available even in developed countries. The scarcity of data on the quality of IRB performance implies unsatisfactory situation of oversight system for human participants research.
In Korea, the enactment of the KGCP (Korean Good Clinical Practice) in 1995 legally mandated clinical trials to be reviewed and continuously monitored by IRBs. Several studies showed that the enforcement of the KGCP has exerted remarkably positive influences on infrastructure and qualities of clinical trials in Korea (2-4). In January 2001, the Korean government revised the KGCP based upon the ICH-GCP (E6) (5). With the revision of the KGCP, legal and institutional bases were established for the constitution and operation of IRBs to be standardized and upgraded to the international level (6).
With their short history, however, the current situation of IRBs in Korea is neither desirable nor satisfactory. Most IRBs confine their roles to review only drug-related research, leaving large area of human participant research untouched by any ethics review. In spite of standardized KGCP regulation, actual IRB practice varies to a great extent from one institution to another, depending on each institution's local situation, experience and resources. While globalization and expansion of clinical trials and biomedical researches call for transparent and competent ethics review, there are no regulations or guidelines for ethics review of biomedical researches except for KGCP for clinical trials in Korea. In this backdrop, in March 2002, IRB members in major hospitals, biomedical researchers, medical directors of pharmaceutical companies and offices from health authorities founded the Korean Association of the Institutional Review Boards (KAIRB) under the Korean Academy of Medical Sciences. The main mission of the KAIRB is to help Korean IRBs build up ethical review capacity to the international level. For this purpose, the KAIRB took initiatives in various activities including co-hosting the Korea-NIH Conference on Ethical and Regulatory Aspects of Human Participant Research in June 2002. Aiming at developing strategies for improving IRBs' ethics review quality based on concrete data, the KAIRB conducted the first nationwide survey to evaluate current situation of IRBs' structure and actual review process.
Based upon the data on IRBs submitted to the Health and Welfare Committee of Korean Congress in 2001, 74 IRBs were included as the correspondents of the nationwide survey. After developing a questionnaire with 67 question items on constitution, review process and institutional policy for research participant protection, we sent it to each IRB's expert whom we identified through call to each IRB as the most knowledgeable person about the IRB. Conducted in April 2002 as the first project of the KAIRB, this survey explicitly notified that the purpose of the survey was to improve quality of ethics review, and all the information from each IRB will be kept confidential to enhance reliability of the data. Of 74 IRBs to which we mailed the questionnaire, 63 returned it in due time (response rate: 63/74=85.1%).
After being reviewed for the completeness and correctness of the information in the returned questionnaires, computerized database was constructed for statistical analysis. The data were presented as proportions in tables and figures because most of the data were categorical variables.
The survey demonstrated that most IRBs in Korea have been established after the implementation of KGCP in 1995. Of 67, there were only 11 IRBs (21%) before the enforcement of the KGCP. Thirty IRBs (57%) were established between 1995-1997, and 9 (17%) between 1998-2001. In most IRBs (91%), the president of the hospital appoints members. In more than two thirds of IRBs, the chairperson of the IRB is appointed by the president (n=29, 46%) or automatically takes the chair (n=19, 30.2%) without having election process within IRB. While 37 (58.7%) IRBs have their own administrators for the efficient IRB operation, 24 (38.1%) have no administrative supports from the institution.
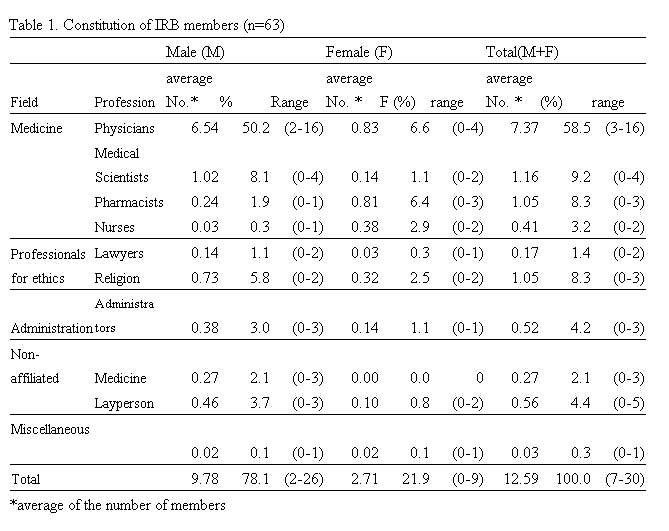
Since sound constitution of IRB members is a prerequisite to proper review, we tried to collect detailed information on the composition of each IRB's membership. The average number of members in an IRB in Korea is 12.6 (range: 7-30). Average percentage of members from medical fields is 79%, among which are physicians (59%; range 3-16), medical scientists (9%; range 0-4), pharmacists (8%; range 0-3) and nurses (3.2%; range 0-2). The average percentage of professionals for ethics review is 10% including professionals from religion (8%; range 0-3) and lawyers (8%; range 0-3). Non-affiliated laypersons were 4.4% (range 0-5) ( Table 1). The gender ratio of the members is 78M/22F. While 90% of IRBs have professionals for ethics review including professionals from religion and lawyers, only 43 % have layperson members. About 40% do not have members who are non-affiliated to the institution.
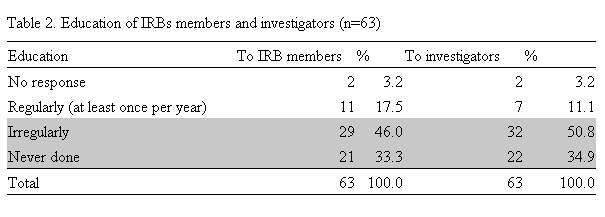
Of 63 IRBs, only 11 provide education to IRB members, and only 7 to investigators on a regular basis at least once per year. Ninety-one percent of IRBs replied that they have written Standard Operating Procedures (SOP) for IRB review, which means 9% breach KGCP (Table 2).
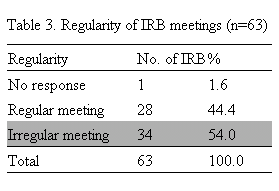
Twenty eight IRBs (44%) hold meetings regularly, while 54% replied that they have irregular meetings (Table 3). Only 19 IRBs (30%) hold meetings once a month or more. These data mean that more than half of all IRBs are not very active.
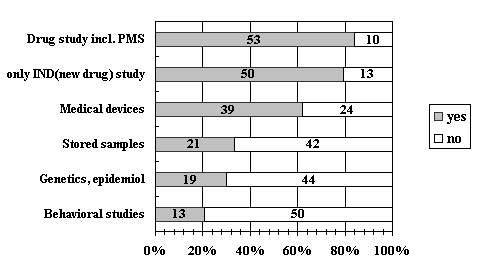
To the question what kinds of research should be reviewed in each IRB, about 30% IRBs make it a rule to review studies on epidemiology or on genetics and 33% IRBs review studies with stored biological samples. Only 21% review behavioral studies. On the other hands, 84% IRB replied that they should review drug studies including Post-marketing surveillance, 79% new drug study, and 62% medical device study (Figure 1).
Figure 1. Type of research to be reviewed in each IRB.
When asked by the number of protocols reviewed in 2001, 24 IRBs (38%) replied that they reviewed 1-5 protocols, 11 (17.5%) reviewed 6-10 protocols and 8 (12.7%) reviewed 11-20 protocols (Table 4). Nine IRBs reviewed more than 50 protocols in 2001. Academic research was reviewed at 19 IRBs (30.2%), of which 2 (3.2%) dealt with more than 10 academic research protocols in 2001.

To the question for whether and how the protocols are thoroughly reviewed before the formal meeting, about 41.3% IRB replied that all the members attend the formal meeting after reading all the protocols. Fourteen percent IRBs adopt primary review system in which the protocols are thoroughly reviewed before the formal meeting by pre-assigned members. About 30% of IRBs, the members do not read protocols before the meeting; some of those IRBs replace the reading with investigators' presentation (Table 5).

Layperson members participate actively in the meeting in 54% of IRBs, understanding protocols to moderate degree. In 54 IRBs (86%), principal investigators are allowed to attend IRB meetings, usually only when asked by IRB (52.4%), while they are not allowed in 6 (9.5%) IRBs. If an IRB member were the investigator of a protocol to be review, the member cannot attend the meeting in 9 IRBs (14.3%), or the member may attend to reply to questions but cannot review in 53 IRBs (80.9%) (Table 6).


Only one IRB allows the member, who is the investigator, to attend and review the protocol. IRB decision is reached by consensus in 36 IRBs (57%), or by majority vote 24 (38%) (Table 7).
Thirty four IRBs (54%) perform continuing review, while 26 IRBs (41%) do not. The patterns of continuing review among IRBs are quite different. Some IRBs have reasonable criteria for the interval of continuing review for each protocol. But the others have fixed review intervals for all the protocols. The interval of the continuing review varies from every three months to once per year (Figure 2).
Figure 2. Continuing review and its intervals

Most IRBs in Korea have expedited review system. Of 56 IRBs (89%) that have expedited review system, 27 IRBs (43%) use subcommittee system or pre-assigned members for expedite review, and 19 (30%) collect opinions from individual members. Immediately reported serious adverse drug reaction is the most important category for expedite review (81%), but categories of expedited review are various among institutions (Table 8).

For the question about review on compensation for the research participants, 46 IRBs (73%) review only for monetary payment, and 7 IRBs (11%) do not review any kinds of compensation (Table 9). Only 22 IRBs (35%) make it obligatory for sponsors to have insurance for indemnity when participants get injured. For academic researches, 35 institutions (62%) do not have any policy for indemnity for research participants (Figure 3). Since non- response rates are high for the questions of payment policy, it is hard to have complete data on the current situation of payment. Institutions have various payment policies, which vary according to the phases of clinical trials (Figure 4). For those who have not completed study, 9-19 % of institutions are allowing for non-payment for the participants.



To the open question asking about problems and difficulties that each IRB faces, each respondent replied one to several items including: 1. Lack of expertise for IRB review; 2. Lack of knowledge and experience on IRB operation; 3. Lack of recognition of the importance of IRB functions; 4. Lack of resources and support from the institute, 5. Lack of active participation of IRB members, 6. Lack of independence.
This study revealed many shortcomings to be improved in the IRB system in Korea. The problems founded through this survey can be summarized into three categories: (1) the structure of ethics review system for human participant research; (2) review process; and (3) policy for protection of research participants.
(1) Limited scope of IRBs review: The most serious problem is that all human related research is not reviewed by IRBs. A very few categories of human researches are covered by IRB review system. Most IRBs limited their review only to legally bounded researches. Researches seeking KFDA approval such as clinical trials for new drugs, biologics, or devices have to undergo IRB review process by the KGCP regulation. Since the KGCP is the only regulation to obligate IRB review, the regulation does not apply to all the other researches that do not seek KFDA approval. The survey result that only 30 % IRBs review academic researches show that there is no ensuring system to protect research participants in academic studies (table 4). Remaining mechanism is the publication requirement of ethics review for human participant researches, which are not effective due to lack of compliance of researchers in Korea. Recently, positive signs appeared for increasing coverage of IRB review. The Stem Cell Research Project supported by the Korean Ministry of Science and Technology, starting October 2002, set internal guidelines that the ethics committee should review all protocols funded by the Project. Researches both socially and ethically controversial such as stem cell research, establishment of the ethics review system will increase transparency of the research practice and thereby public assurance.
(2) Absence of IRBs in institutions other than hospitals: More than half of IRBs in Korea were established in the hospitals accredited in 1993 for clinical trials right before the implementation of KGCP in 1995. Many other institutions conducting human participant researches or the equivalent researches such as universities or biomedical research centers do not have IRBs. This reflects the structural problems that currently many categories of academic researches are not reviewed by IRB.
(3) Lack of independence of IRBs: Most IRBs have the problem of lack of independence. The heart of ethics review is the IRB's independence from political, institutional, professional and market influences in their composition, procedures and decision-making.(7) In 91% IRBs, the president of the hospital appoints members, and in more than two thirds of IRBs, the chairperson is appointed by the president (n=29, 46%) or automatically takes the chair (n=19, 30.2%). As Emanuel pointed out, the IRB has inherent institutional conflicts of interest because most IRB members are working for the very institution conducting the research they review.(8) It makes the IRB and individual members inclined influenced by the senior officials and the peer relationship with researchers within the institution.
(4) Improper balance in constituting IRB members: For conducting competent and independent review, it is crucial to have sound composition of IRB members. It should include relevant scientific and ethical expertise and laypersons, with balanced professions, age and gender distribution.(8) Our survey shows that except male physicians (50% of all members) the constitution does not reflect diversity in terms of representation of professions and genders. Most serious problems are that only 43 % have layperson members and about 40% do not have members who are non-affiliated to the institute. This may lead to weakening independence of the IRB and to depriving awareness of the community.
(5) Inadequate education of IRB members and investigators: Although the KGCP requires regular education to IRB members and clinical investigators, our survey shows that less than 20 % institutions provide regular education to IRB members and about 10 % to investigators (Table 2). Some institutes provide annual workshop programs and courses on human research for researchers and IRB members. And yet, there is no specified standard curriculum available for training ethical research practice even in these institutions.
(1) Variable quality and expertise of IRB reviews: There are significant variability in quality and expertise in IRB reviews. A few IRBs have members more experienced and diverse expertise for reviewing various kinds of protocols, while rest do not. Review practice itself is various among IRBs. While all members read and review documents before the meeting in 41% IRBs, for example, document review is replaced by investigators' presentation in 14% IRBs. As the respondent replied (Figure 5), lack of experience and lack of expertise for IRB review and operation are most serious problems in many IRBs.
(2) Insufficient continuing review: While the KGCP revised in 2001 addresses continuing review, only half of responded IRBs conduct continuing review. Since the questionnaire was not in detail enough to gather information on how they conduct continuing review, we cannot tell how effectively and efficiently those continuing reviews are conducted as an assuring system to protect research participants. As the concept of continuing review was recently introduced to the KGCP in 2001, several IRBs have been gearing toward preparation of continuing review with electronic database system.
(3) Variable expedited review: While most IRBs in Korea have expedited review system to deal with reports on serious adverse events or minor changes of the protocols. About 10 %, however, do not have expedited review system, which may cause time-consuming, inefficient review process. The categories of expedite review are variable. Even 40 % IRBs use expedite review system to review amendment of protocols that may increase risk to participants, or exert serious impact to the clinical trial, which should be reviewed regular IRB meeting with full members. These data (Table 8) suggest that specific guidelines on expedite review should be prepared.
(1) Deficiency of review of compensation for research participants: One of the most important functions of the IRB is to ensure the protection of research participants, including reviewing and overseeing the safety measures for the participants. Although international guidelines suggest reviewing all kinds of compensation such as treatment, prorated payment, indemnity, gift, and other benefits. Survey shows most IRBs review only for monetary payment. About 10 % IRBs do not review any compensations for the participants.
(2) Variable policies for protection of participants in clinical trial: In case of clinical trial, about 50% of institutions make sponsors indemnify for juries or other harms of participants. Only 22 IRBs (35%) make it obligatory for sponsors to have insurance for indemnity when participants get injured. Except 3 institutions that have no policy, most have some kinds of policy for indemnity for the participant in clinical trial mainly because sponsors provide the cost by any means.
(3) Lack of policies for protection of participants in academic research: More serious than in the case of clinical trials is the lack of policies for protection of participants in academic researches. Currently, this becomes obvious and more serious, as more researchers involving academic researches face the problem. The financial resources for indemnity for participants in academic researches are not as clear as in clinical trials. Usually, research funds come from the government or public research institutes, which usually have tight budget for researches. Two-thirds of institutions do not have any policy for indemnity for research participants. In 21% institutions, principal investigators have to pay for indemnity; in 10%, institutions have responsibility for it. So far insurance system for academic researches has not developed, and problems will be growing.
In spite of several problems in the IRB system discussed above, this data is by no means complete or with no error. Since this study relied solely on the reply of the expert member of each IRB to the questionnaire, further evaluation by an independent thirty party is necessary for the systematic collection of thorough and valid data.
To improve current situation of human research participant protection system in Korea, the authors make following suggestions:
1. Enactment for IRB reviews: In spite of the explosion of human resources researches in Korea, it has only a regulation for researches seeking KFDA approval (KGCP). In order to provide public assurance, it is necessary to make regulations to obligate other human research categories to be reviewed by for research ethics review committees. At least, government-funded researches that virtually citizen and taxpayers finance should be reviewed by IRBs to ensure the protection of research participants.
2. Building and strengthening networks for quality improvement: Many respondents wrote that individual IBR suffers from lack of experiences, knowledge, expertise, and resources. To improve quality of IRB review, it is important to build networks at regional, national and international levels to exchange information, utilize resources efficiently and standardize quality of review. The Korean Association of IRBs (KAIRB) is expected to play important roles in supporting local IRBs by regularly providing necessary information and educating IRB members. The KARIB is recently developed a general guideline for constituting and operating IRBs to encompass hitherto uncovered areas of human participants researches. There is also plenty room to organize meetings of regional IRBs in Korea including joint IRBs for multi-center researches. At the international level, WHO is organizing regional forums of ethics committees that review biomedical researches. Based upon their analysis of the problem of human subject research protection, FDA and OHRP of U.S.A. already geared up toward building capacities of ethics review committees in developing countries in collaboration with WHO.(9)
3. Training and education: For quality improvement of IRB review, continuing education and training is essential. As several important reports as well as our respondents of the survey pointed out, one of the serious problems of IRBs in developing countries including Korea is lack of expertise in IRBs. Primarily individual institution ought to take responsibility for training of the IRB members and the investigators in the ethics of human participants research. Realistically speaking, when the lack of resources of an individual institute is a real problem, the KAIRB, relevant academic societies, FDA or other responsible governmental organizations should develop and provide curricula and course programs including basic, more advanced, and specific courses for all IRB members and investigators.
4. Development of quality assurance system (accreditation): Accreditation of a research institution means that the institution fulfills certain predetermined standards of human participants protections. At this moment, IRBs in Korea need to strive for quality improvement, and introduction of accreditation seems too far from the reality. Since more researchers' recognize the importance of IRB review, however, it is timely to consider the accreditation system tying together with IRB's continuing process of self-assessment, self-improvement and auditing. With globalization of biomedical researches, international agencies' oversight activities over IRBs will increase through auditing or inspection (9-12). As a part of peer review, or for the educational purpose, auditing or surveying IRBs can be introduced in Korea. After all, these activities will be Korean IRBs' continuous efforts to build capacity for conducting biomedical researches with highest attainable quality in science and ethics.
(1) The World Medical Association. Declaration of Helsinki. Revised at 52nd WMA General Assembly, Edinburgh, Scotland, October 2000.
(2) Shin SG. Current status of clinical trials and GCP in the Republic of Korea. Drug Inf J 1997; 31: 1079-1087.
(3) Shin SG. The current status of clinical trials in the Republic of Korea. Drug Inf J 1998; 32: 1217S-1222S.
(4) Howard L, Charles K, Shin SG. Changes in clinical trial practice and the working environment in the Korean pharmaceutical industry since the implementation of Good Clinical Practice. Drug Inf J 2001; 35: 203-210.
(5) ICH Harmonized Tripartite Guideline: E6. Good Clinical Practice: Consolidated Guideline. May 1996.
(6) Korean Ministry of Health and Welfare. Guidelines of Korean Good Clinical Practice Guidelines. January 2001.
(7) World Health Organization. Operational Guidelines for Ethics Committees That Review Biomedical Research. Geneva: WHO, 2000.
(8) Wood A, Grady C, Ezekiel J. The Crisis in Human Participants Research: Identifying the Problems and Proposing Solutions. This paper was first presented and discussed at the meeting of U.S.A. President's Council on Bioethics September 12, 2002. http://www.bioethics.gov/emanuelpaper.html
(9) Office of the Inspector General, Department of Health and Human Services. The Globalization of Clinical Trials: A Growing Challenge in Protecting Human Subjects. September 2001. OEI-01-00-00190.
(10) World Health Organization. Surveying and Evaluating Ethical Review Practices, a companion guideline to the TDR WHO Operational Guidelines for Ethics Committees That Review Biomedical Research (2000). Geneva: WHO, 2002.
(11) European Forum for Good Clinical Practice. European Guidelines for Auditing Independent Ethics Committees. Brussels: EFGCP, 2002.
(12) European Network of GCP Auditors and Other GCP Experts. The ENGAGE Guideline: Optional Guideline for Good Clinical Practice Compliance and Quality Systems Auditing. Salve 1. Supplement to The EFGCP News, September 1998.
1 This work was supported by the Science Culture Research Center (SCRC) Grant in 2002.